A Challenge to Gene Theory,
a Tougher Look at Biotech
by Denise Caruso on 1 July 2007 in the New York Times
THE $73.5 billion global biotech business may soon have to grapple with a discovery that calls into question the scientific principles on which it was founded.
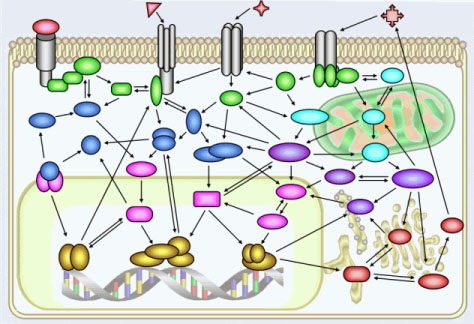
Last month, a consortium of scientists published findings that challenge the traditional view of how genes function. The exhaustive four-year effort was organized by the United States National Human Genome Research Institute and carried out by 35 groups from 80 organizations around the world. To their surprise, researchers found that the human genome might not be a “tidy collection of independent genes” after all, with each sequence of DNA linked to a
single function, such as a predisposition to diabetes or heart disease.
Instead, genes appear to operate in a complex network, and interact and overlap with one another and with other components in ways not yet fully understood. According to the institute, these findings will challenge scientists “to rethink some long-held views about what genes are and what they do.”
Biologists have recorded these network effects for many years in other organisms. But in the world of science, discoveries often do not become part of mainstream thought until they are linked to humans.
With that link now in place, the report is likely to have repercussions far beyond the laboratory. The presumption that genes operate independently has been institutionalized since 1976, when the first biotech company was founded. In fact, it is the economic and regulatory foundation on which the entire biotechnology industry is built.
Innovation begets risk, almost by definition. When something is truly new, only so much can be predicted about how it will play out. Proponents of a discovery often see and believe only in the benefits it will deliver. But when it comes to innovations in food and medicine, belief can be dangerous. Often, new information is discovered that invalidates the principles — thus the claims of benefit and, sometimes, safety — on which proponents have built their
products.
For example, antibiotics were once considered miracle drugs that, for the first time in history, greatly reduced the probability that people would die from common bacterial infections. But doctors did not yet know that the genetic material responsible for conferring antibiotic resistance moves easily between different species of bacteria. Overprescribing antibiotics for virtually every ailment has given rise to “superbugs” that are now virtually unkillable.
The principle that gave rise to the biotech industry promised benefits that were equally compelling. Known as the Central Dogma of molecular biology, it stated that each gene in living organisms, from humans to bacteria, carries the information needed to construct one protein.
Proteins are the cogs and the motors that drive the function of cells and, ultimately, organisms. In the 1960s, scientists discovered that a gene that produces one type of protein in one organism would produce a remarkably similar protein in another. The similarity between the insulin produced by humans and by pigs is what once made pig insulin a life-saving treatment for diabetics.
The scientists who invented recombinant DNA in 1973 built their innovation on this mechanistic, “one gene, one protein” principle.
Because donor genes could be associated with specific functions, with discrete properties and clear boundaries, scientists then believed that a gene from any organism could fit neatly and predictably into a larger design — one that products and companies could be built around, and that could be protected by intellectual-property laws.
This presumption, now disputed, is what one molecular biologist calls “the industrial gene.”
“The industrial gene is one that can be defined, owned, tracked, proven acceptably safe, proven to have uniform effect, sold and recalled,” said Jack Heinemann, a professor of molecular biology in the School of Biological Sciences at the University of Canterbury in New Zealand and director of its Center for Integrated Research in Biosafety.
In the United States, the Patent and Trademark Office allows genes to be patented on the basis of this uniform effect or function. In fact, it defines a gene in these terms, as an ordered sequence of DNA “that encodes a specific functional product.”
In 2005, a study showed that more than 4,000 human genes had already been patented in the United States alone. And this is but a small fraction of the total number of patented plant, animal and microbial genes.
In the context of the consortium’s findings, this definition now raises some fundamental questions about the defensibility of those patents.
If genes are only one component of how a genome functions, for example, will infringement claims be subject to dispute when another crucial component of the network is claimed by someone else? Might owners of gene patents also find themselves liable for unintended collateral damage caused by the network effects of the genes they own?
And, just as important, will these not-yet-understood components of gene function tarnish the appeal of the market for biotech investors, who prefer their intellectual property claims to be unambiguous and indisputable?
While no one has yet challenged the legal basis for gene patents, the biotech industry itself has long since acknowledged the science behind the question.
“The genome is enormously complex, and the only thing we can say about it with certainty is how much more we have left to learn,” wrote Barbara A. Caulfield, executive vice president and general counsel at the biotech pioneer Affymetrix, in a 2002 article on Law.com called “Why We Hate Gene Patents.”
“We’re learning that many diseases are caused not by the action of single genes, but by the interplay among multiple genes,” Ms. Caulfield said. She noted that just before she wrote her article, “scientists announced that they had decoded the genetic structures of one of the most virulent forms of malaria and that it may involve interactions among as many as 500 genes.”
Even more important than patent laws are safety issues raised by the consortium’s findings. Evidence of a networked genome shatters the scientific basis for virtually every official risk assessment of today’s commercial biotech products, from genetically engineered crops to pharmaceuticals.
“The real worry for us has always been that the commercial agenda for biotech may be premature, based on what we have long known was an incomplete understanding of genetics,” said Professor Heinemann, who writes and teaches extensively on biosafety issues.
“Because gene patents and the genetic engineering process itself are both defined in terms of genes acting independently,” he said, “regulators may be unaware of the potential impacts arising from these network effects.”|
Yet to date, every attempt to challenge safety claims for biotech products has been categorically dismissed, or derided as unscientific. A 2004 round table on the safety of biotech food, sponsored by the Pew Initiative on Food and Biotechnology, provided a typical example:
“Both theory and experience confirm the extraordinary predictability and safety of gene-splicing technology and its products,” said Dr. Henry I. Miller, a fellow at the Hoover Institution who represented the pro-biotech position. Dr. Miller was the founding director of the Office of Biotechnology at the Food and Drug Administration, and presided over the approval of the first biotech food in 1992.
Now that the consortium’s findings have cast the validity of that theory into question, it may be time for the biotech industry to re-examine the more subtle effects of its products, and to share what it knows about them with regulators and other scientists.
This is not the first time it has been asked to do so. A 2004 editorial in the journal Nature Genetics beseeched academic and corporate researchers to start releasing their proprietary data to reviewers, so it might receive the kind of scrutiny required of credible science.
According to Professor Heinemann, many biotech companies already conduct detailed genetic studies of their products that profile the expression of proteins and other elements. But they are not required to report most of this data to regulators, so they do not. Thus vast stores of important research information sit idle.
“Something that is front and center in the biosafety community in New Zealand now is whether companies should be required to submit their gene-profiling data for hazard identification,” Professor Heinemann said. With no such reporting requirements, companies and regulators alike will continue to “blind themselves to network effects,” he said.
The Nature Genetics editorial, titled “Good Citizenship, or Good Business?,” presented its argument as a choice for the industry to make. Given the significance of these new findings, it is a distinction without a difference. |